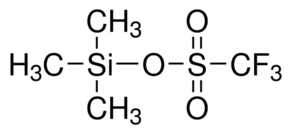
Trimethylsilyl Trifluoromethanesulfonate
Product Details:
- Usage As a silylating agent for the synthesis of trimethylsilyl-enol ethers from esters of -diazoacetoacetic acid. To activate benzyl and allyl ethers for the alkylation of sulfides. To facilitate the conversion of Diels-Alder adducts of Danishefskys diene to cyclohexenones without the formation of methoxy ketone by-product. To prepare difluoroboron triflate etherate, a powerful Lewis acid especially in acetonitrile solvent. As a reagent in a Dieckmann-like cyclization of ester-imides and diesters.
- Melting Point 77 C/80 mmHg (lit.)
- Molecular Formula C4H9F3O3SSi
- Refractive Rate n20/D 1.36 (lit.)
- Solubility Miscible with aliphatic, aromatic hydrocarbons, haloalkanes and ethers. Immiscible with water.
- Molecular Weight 222.26 Grams (g)
- Application Organic Synthesis
- Click to view more
X
Trimethylsilyl Trifluoromethanesulfonate Product Specifications
- n20/D 1.36 (lit.)
- C4H9F3O3SSi
- 77 C/80 mmHg (lit.)
- As a silylating agent for the synthesis of trimethylsilyl-enol ethers from esters of -diazoacetoacetic acid. To activate benzyl and allyl ethers for the alkylation of sulfides. To facilitate the conversion of Diels-Alder adducts of Danishefskys diene to cyclohexenones without the formation of methoxy ketone by-product. To prepare difluoroboron triflate etherate, a powerful Lewis acid especially in acetonitrile solvent. As a reagent in a Dieckmann-like cyclization of ester-imides and diesters.
- Organic Synthesis
- 222.26 Grams (g)
- Miscible with aliphatic, aromatic hydrocarbons, haloalkanes and ethers. Immiscible with water.
- 248-565-4
- >98%
- Liquid
- 27607-77-8
- 29033919
- Other
- C4H9F3O3SSi
- Clear colorless to slight yellow liquid
- 2 Years
- Organic Compound
- 1.225 g/mL Gram per millilitre (g/mL)
Trimethylsilyl Trifluoromethanesulfonate Trade Information
- Ankleshwar
- Cash in Advance (CID) Cash Advance (CA)
- Contact us for information regarding our sample policy
- GLASS BOTTLES
- Australia South America Western Europe Middle East Africa Central America Asia Eastern Europe North America
- All India
- ISO
Product Description
Trimethylsilyl Trifluoromethanesulfonate is functional as a trifluoromethanesulfonate derivate belonging to the group of trimethylsilyl R. It has kindred reactivity to trimethylsilyl chloride. It is rendered with appreciable shelf life as well as increased effectiveness. Applicable in chemical as well as medical industry the utilization of this chemical needs proper recommendation as well as accurate amount. Offered Trimethylsilyltrifluoromethanesulfonate is employed with special chemical properties as well as accurate pH value.
Enter Buying Requirement Details
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese