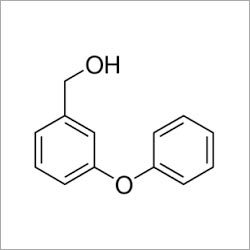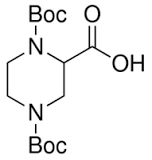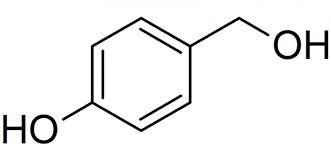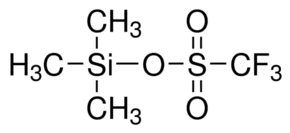
TMS Triflate
Product Details:
Product Description
Tms Triflate is an excellent leaving group that can be employed in certain organic reactions such as Suzuki couplings and Heck reactions and nucleophilic substitution. Since alkyl triflates are enormously reactive in SN2 reactions, it must be stored in states free of nucleophiles(such as water). It is offered with stability to resonance stabilization that reasons the negative charge to be extent over the three oxygen atoms as well as the sulfur atom. It can enable additional stabilization by working as a strong electron-withdrawing group. Offered Tms Triflate has been employed in Takahashi Taxol total synthesis as well as chemical glycosylation reactions. It is also applicable to deprotect silyl enol ethers, by modifying them into aldehydes or ketones.
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese