Trifluoromethanesulfonic Acid
Product Details:
X
Product Description
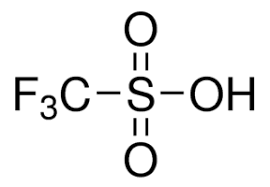
Trifluoromethanesulfonic Acid is considered to be one of the strongest acids and finds application as catalyst for esterification. It has molecular formula CF3SO3H with molar mass of 150.08 g/mol. This compound appears as colorless liquid with density of 1.696 g/ml and boiling point of about 162 degrees Celsius. It is miscible in water and shows melting point of -40 degrees Celsius. Trifluoromethanesulfonic Acid can industrially be produced by electrochemical fluorination of methanesulfonic acid under specific conditions. It is also known to have hygroscopic property and shows solubility in polar solvents. Also known as sulfonic acid, this compound appears as viscous liquid with high purity level.
Trifluoromethanesulfonic Acid Properties:
1. Acidity:
- TfOH is a strong acid and is considered a superacid, meaning it has acidity much higher than conventional mineral acids like sulfuric acid or hydrochloric acid.
- It has a very low pKa value, indicating a high degree of ionization in solution.
2. Solubility:
- Trifluoromethanesulfonic acid is highly soluble in polar solvents, such as water, due to its strong polar nature.
3. Reactivity:
- TfOH is a versatile catalyst in various chemical reactions, particularly in organic synthesis.
- It is often used as a catalyst in reactions like Friedel-Crafts acylation and alkylation, isomerization, esterification, and other acid-catalyzed processes.
4. Corrosiveness:
- Due to its strong acidity, trifluoromethanesulfonic acid can be corrosive to certain materials, and care must be taken in handling and storage.
- It can react with some metals and may corrode certain types of containers.
5. Hygroscopicity:
- TfOH is hygroscopic, meaning it readily absorbs moisture from the air. This property can affect its concentration and reactivity, so it is often stored under anhydrous conditions.
Trifluoromethanesulfonic Acid Applications:
1. Organic Synthesis: TfOH is widely used as a strong acid catalyst in organic synthesis. It is employed in reactions such as Friedel-Crafts acylation and alkylation, esterification, cyclization, and isomerization.
2. Polymerization Reactions: It serves as a catalyst in certain polymerization reactions, including the polymerization of olefins and the synthesis of specialty polymers.
3. Dehydration Reactions: TfOH is utilized in dehydration reactions, where it helps remove water from a reaction mixture, promoting the formation of desired products.
4. Alkylation of Aromatics: Trifluoromethanesulfonic acid is employed in the alkylation of aromatics, a process used to introduce alkyl groups into aromatic compounds.
5. Esterification: It facilitates esterification reactions, converting carboxylic acids and alcohols into esters.
6. Deacetylation Reactions: TfOH can be used for deacetylation reactions, removing acetyl groups from organic compounds.
7. Isomerization: It is involved in isomerization reactions, where it facilitates the conversion of one isomer into another.
8. Catalysis in Chemical Transformations: TfOH is applied in various chemical transformations where a strong acid catalyst is required to drive the reaction to completion.
9. Electrochemistry: In some electrochemical processes, trifluoromethanesulfonic acid is used as an electrolyte.
10. Fuel Cells: It may find applications in proton exchange membrane fuel cells (PEMFCs) as a component in the electrolyte system.
11. Pharmaceutical Industry: TfOH is employed in the synthesis of pharmaceutical intermediates and in the production of specific drugs.
12. Analytical Chemistry: Trifluoromethanesulfonic acid can be used in analytical techniques, such as high-performance liquid chromatography (HPLC).
FAQs of Trifluoromethanesulfonic Acid:
Q: What is the boiling point of Trifluoromethanesulfonic Acid?
A: The boiling point of Trifluoromethanesulfonic Acid is 162 C.Q: What is the molecular weight of Trifluoromethanesulfonic Acid?
A: The molecular weight of Trifluoromethanesulfonic Acid is 150.07121 g/mol.Q: What is the storage recommendation for Trifluoromethanesulfonic Acid?
A: Trifluoromethanesulfonic Acid should be stored at room temperature.Q: What is the grade of Trifluoromethanesulfonic Acid?
A: Trifluoromethanesulfonic Acid is available in medicine grade and reagent grade.Q: What is the application of Trifluoromethanesulfonic Acid?
A: Trifluoromethanesulfonic Acid is generally a very good solvent for organic compounds that are capable of acting as proton acceptors in the medium.Enter Buying Requirement Details
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese